
Raw and excipients compatibility
Screening of formulation and research of preparation process(Such as emulsification and sterilization process)
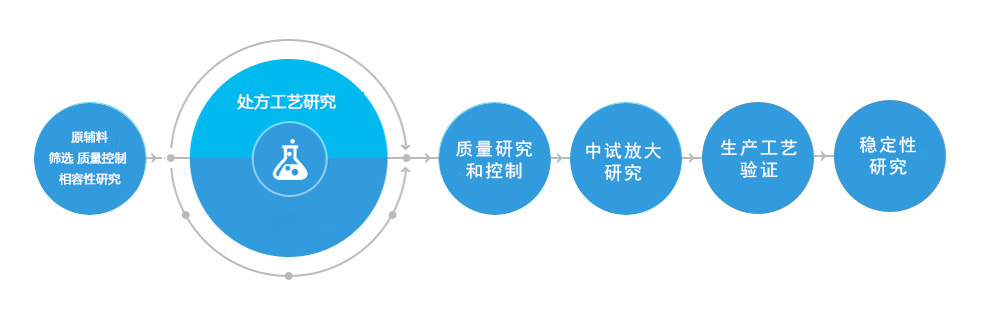

Raw material selection and internal control
Research and establishment of analytical methods(such as assay and related substances)
Study of physical and chemical indexes(such as size(macroparticle), anisidine value, peroxide value, free fatty acid, lysophospholipid, etc.)

Compatible stability
Stress tests
Accelerated, long-term stability test
 FBC
FBC
Copyright © 2019 FBC (Shanghai) Pharmaceutical Technology Co., LTD. All Rights Reserved
备案号:沪ICP备16038274号-1